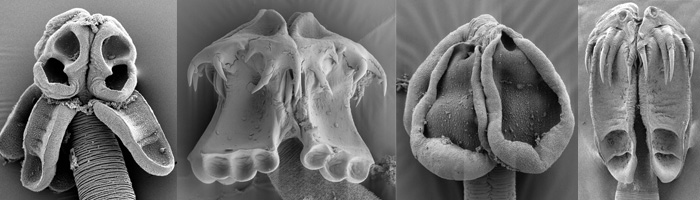
Scanning electron micrographs of Rhoptrobothrium myliobatidis, Triloculatum bullardi, Paraorygmatobothrium sp., Acanthobothrium sp.
Scolex normally possessing 4 muscular bothridia that are widely variable in form. Bothridia sessile or occassionally stalked, with or without hooks. In some groups left and right pairs of bothridia are arranged back-to-back, each pair being born on a pedicel. Proglottids generally hermaphroditic (except Dioecotaenia); most euapolytic, some apolytic or hyperapolytic. Multiple testes present. Vagina opens anterior to cirrus-sac. Genital pores usually lateral, occasionally sublateral. Ovary posterior, bilobed or tetralobed in cross section. Vitellarium follicular; follicles arranged in lateral fields (or infrequently circumcortical). Uterus tubular to sacciform, occasionally with lateral branches; pre-formed uterine pores lacking.
Over 400 species, and 64 genera recognized as valid; familial classification extremely uncertain.
The order is almost certainly NOT monophyletic, but may include subsets of monophyletic taxa. Closest relatives of some tetraphyllideans may include the Proteocephalidea; in other cases the Lecanicephalidea or Cyclophyllidea. Several groups recognized as tetraphyllideans by some previous workers are generally now considered to represent independent orders (e.g., Litobothriidea, Cathetocephalidea, Rhinebothridea).
Elasmobranchs (i.e., sharks and rays); essentially all elasmobranch species examined to date are known to host 1 or more species of tetraphyllideans; exceptional is Chimaerocestus, which parasitizes the sister-group of elasmobranchs, the Holocephala (chimaeras).
Spiral intestine.
Cosmopolitan, with majority of species known from tropical and subtropical waters.
No complete life-cycle is known. Larval stages likely include hexacanth embryo, procercoid, followed by a plerocercoid or merocercoid as the terminal larval stage. Most likely possess a 3-5 host life-cycle. Larval hosts include teleosts, mollusks, crustaceans, and marine mammals. Substantial progress has recently been made using molecular markers (see Jensen and Bullard, 2010).
Selected References:
Euzet, L. 1956. Recherches sur les cestodes tetraphyllides des selaciens côtes de France. Thesis. Université de Montpellier, 263 pp. PDF
Euzet, L. 1994. Order Tetrapyllidea. pp. 149-194. In Khalil, L. F., Jones, A., Bray, R. A. (Eds.) Keys to the cestode parasites of vertebrates. CAB International, Wallingford, U.K. PDF
Caira, J. N., K. Jensen, and C. J. Healy. 1999. On the phylogenetic relationships among the tetraphyllidean, lecanicephalidean and diphyllidean tapeworm genera. Systematic Parasitology 42: 77-151. PDF
Caira, J. N., K. Jensen, and C. J. Healy. 2001. Interrelationships among tetraphyllidean and lecanicephalidean cestodes. pp. 135-158. In Littlewood, D. T .J., Bray, R. (Eds.) Interrelationships of the Platyhelminthes, Taylor and Francis. PDF
Jensen, K. and S. A. Bullard. 2010. Characterization of a diversity of tetraphyllidean and rhinebothriidean cestode larval types, with comments on host associations and life-cycles. International Journal for Parasitology. 40: 889–910. PDF
Ruhnke, T. R. 2010. A monograph on the Phyllobothriidae (Platyhelminthes: Cestoda). Bulletin of the Nebraska State Museum. 25: 205 pp. PDF
Taxon Coordinator:
Dr. Janine N. Caira
University of Connecticut
Department of Ecology and Evolutionary Biology
75 N. Eagleville Rd., Unit 3043
Storrs CT 06269-3043
U.S.A.
E-MAIL: janine.caira@uconn.edu

